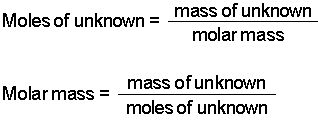
Note: Although Na 2CO 3 is an ionic compound and sodium is present in an ionic form, it is a habitual way of referring to an element in these types of calculations. This is the accurate way of rounding off to the correct number of significant figures in multistep calculations. I do it here mainly to save space, but you can keep the numbers in the calculator and round them off at the end. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. The little inconsistency in the final answer is due to rounding off the intermediate numbers. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. So, to calculate the moles from a given mass, we make a conversion factor correlating 1 mole with the molar of the given component.įor example, how many moles of sulfur are there in a 16.2 g sample? The molar mass, on the other hand, is a constant number for a given atom or a molecule as it is for a specific amount of it. 10.5 kg of Na 2 SO 4 ♱0H 2 O in 18.60 L of solution. 0.2074 g of calcium hydroxide, Ca (OH) 2, in 40.00 mL of solution. 98.0 g of phosphoric acid, H 3 PO 4, in 1.00 L of solution. The given mass is the mass of the sample, and it can be any number, for example, we can have 10 g of salt, 15 g, or 100 g. Determine the molarity for each of the following solutions: 0.444 mol of CoCl 2 in 0.654 L of solution. Lets practice determining the molar mass of some common substances. The periodic table lists the molar mass of each element. To calculate the moles from a given mass (m), the molar mass of the component is used. Avogadros number is 6.02 x 10 23 atoms per mole or molecules per mole. The molecular mass of water is 18.0 amu, and the molar mass is 18.0 g/mol.įor the molar mass, keeping one decimal is usually an acceptable approximation, and for the Avogadro’s number, you can use 6.02 x 10 23. Numerically, the molar mass is equal to the atomic mass of a given atom or a molecule, so we can look up the molar mass of an element in the periodic table.įor example, we can say that the mass of Cu is 63.55 amu or 63.55 g/mol. The mass of one mole of atoms/molecules/ions is called its molar mass (M) expressed in g/mol. One mole of any element contains 6.022 x 10 23 of its atoms, and a mole of a compound contains 6.022 x 10 23 molecules, ions-ions, and in general, 1 mol = 6.022 x 10 23 particles. This is called Avogadro’s number ( N A = 6.022 x 10 23). By definition, a mole is the number of atoms in a 12-g sample of 12C isotope which happens to be 6.022 x 10 23 atoms. The mole is just a number like a dozen, a hundred, or a million.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
